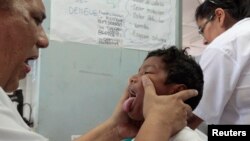
The French drug manufacturer Sanofi Pasteur says its vaccine against dengue fever protected more than half of all children in a late-stage clinical trial. Dengue fever is a mosquito-borne illness that infects an estimated 400 million people each year.
The vaccine was tested in Indonesia, Malaysia, the Philippines, Thailand and Vietnam on more than 10,000 children, protecting 56 percent of them. Another large trial is underway in Latin America. Those results are expected later this year.
Experts say a 50 percent reduction in dengue cases is significant. Olivier Charmeil, chief executive of Sanofi Pasteur, is quoted as saying, "Our goal is to make dengue the next vaccine-preventable disease."
The U.S. Centers for Disease Control and Prevention (CDC) estimates that one-third of the world's population living in tropical and subtropical regions is at risk for illness and death caused by dengue fever, sometimes called "breakbone fever."
Symptoms of infection, include a sudden high fever, severe headache, eye pain, joint pain and pain in muscles and bones. Some people also develop a rash, gum and nose bleeding as well as easy bruising. Currently, there are no vaccines to prevent the viral infection, which is spread by mosquitoes.
When the infection is recognized early, the only treatment for dengue fever is prompt, supportive care, which can substantially reduce the risk of complications and death. Otherwise, people are told to avoid mosquitoes.
The disease was first recognized as a worldwide problem in the 1950s, and the hunt for a successful vaccine has been on ever since then. Among other agencies, Sanofi worked with the U.S. Army on its vaccine. Dengue fever is endemic to Latin America, Southeast Asia and the Pacific Islands.
The vaccine was tested in Indonesia, Malaysia, the Philippines, Thailand and Vietnam on more than 10,000 children, protecting 56 percent of them. Another large trial is underway in Latin America. Those results are expected later this year.
Experts say a 50 percent reduction in dengue cases is significant. Olivier Charmeil, chief executive of Sanofi Pasteur, is quoted as saying, "Our goal is to make dengue the next vaccine-preventable disease."
The U.S. Centers for Disease Control and Prevention (CDC) estimates that one-third of the world's population living in tropical and subtropical regions is at risk for illness and death caused by dengue fever, sometimes called "breakbone fever."
Symptoms of infection, include a sudden high fever, severe headache, eye pain, joint pain and pain in muscles and bones. Some people also develop a rash, gum and nose bleeding as well as easy bruising. Currently, there are no vaccines to prevent the viral infection, which is spread by mosquitoes.
When the infection is recognized early, the only treatment for dengue fever is prompt, supportive care, which can substantially reduce the risk of complications and death. Otherwise, people are told to avoid mosquitoes.
The disease was first recognized as a worldwide problem in the 1950s, and the hunt for a successful vaccine has been on ever since then. Among other agencies, Sanofi worked with the U.S. Army on its vaccine. Dengue fever is endemic to Latin America, Southeast Asia and the Pacific Islands.