India has granted emergency authorization to two vaccines for COVID-19, setting the stage for rolling out what could be the world’s largest immunization program.
The two vaccines are the British-developed AstraZeneca vaccine that is being produced in India and another developed by an Indian pharmaceutical company, Bharat Biotech.
In a brief statement, the Drugs Controller General of India, Venugopal G. Somani said that “the vaccines have been approved for restricted use in India.” He said both manufacturers had submitted data showing their vaccines were safe to use.
India’s green signal to the AstraZeneca vaccines comes within days of approval granted in Britain. An Indian company, the Serum Institute of India has already stockpiled 50 million doses of the vaccine that it made even before formal approvals had come.
Prime Minister Narendra Modi called the vaccine approvals “a decisive turning point to strengthen a spirited fight.” He tweeted that "It would make every Indian proud that the two vaccines that have been given emergency use approval are made in India!"
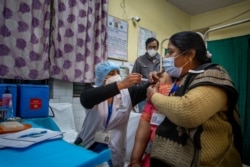
India, the world’s second worst-hit nation, plans to launch its immunization program sometime soon this month and has set the ambitious target of immunizing 300 million people by August. These will include health care workers, security personnel, those over 50 years and the clinically vulnerable.
Reaching that goal will involve administering 600 million doses – the vaccines require to be taken in two doses.
In recent weeks, authorities have raced to prepare cold storage facilities and have trained thousands of healthcare workers to administer the shots. The delivery system was tested with a nationwide “dry run” on Saturday that involved administering dummy shots.
“The logistics have been worked out with the different state governments,” said K. Srinath Reddy, President of the Public Health Foundation of India. But he points out that reaching the target of 300 million will “depend of course on the availability of the vaccine in terms of the volume and in terms of the health workforce that is available.”
The 50 million doses of the AstraZeneca vaccine already available in the country give India an advantage. The Serum Institute, which is the world’s largest vaccine producer says it is on track to ramp up production significantly this month. After the approval, its chief executive Adar Poonawalla tweeted that all the risks it took “with stockpiling the vaccine have finally paid off.”
Experts are optimistic that the vaccines will afford protection even from the mutant strain from Britain that is highly transmissible – nearly 30 cases have been detected raising worries at a time when the daily count of new infections is on a steep downward trend.
“The mutant virus will not be able to evade the vaccine-induced immune mechanisms so it is unlikely that there will be any significant problem over there,” said Reddy.
Some medical experts have raised questions over lack of adequate data on the vaccine developed by Bharat Biotech that is still conducting stage three trials.
Somani said that it was “safe” and has been approved "in public interest as an abundant precaution, in clinical trial mode, to have more options for vaccinations, especially in case of infection by mutant strains."
The approval of the vaccines brings hope to a country where the virus has infected over 10 million and claimed nearly 150,000 lives.
Experts are also giving reassuring messages to those worried about the possible side effects of vaccines that have been developed more quickly than at any time in the past.
“It does protect you against severe illness and that is going to be very important in terms of lifesaving,” says Reddy. And in a country where many young people have had mild infections, he points out that “even younger people have now been known, even if they do not manifest many symptoms, to suffer silent damage to their lungs and heart.”
Both the vaccines approved for India can be stored in ordinary refrigerators at two to eight degrees Celsius. They are also more affordable than the Pfizer and Moderna vaccines – important in a country of 1.3 billion that will need to vaccinate substantial numbers to control the pandemic in the world’s second worst hit nation.